Upon returning home from a Colorado ski trip in late February, the Carl family experienced flu-like symptoms: a fever, shortness of breath, dry cough, and intense fatigue.
At the time, only a handful of Canadians had tested positive for the novel coronavirus and Colorado wouldn’t report its first cases until early March. But Richard Carl said they’d read a news article about a Canadian woman who had returned from a trip to the same area, and tested positive.
“Out of an abundance of caution” a family doctor said to get tested for COVID-19, Carl, 64, told HuffPost Canada. He, his wife and two sons all tested positive, becoming among the first reported cases in the country.
Fortunately, Carl said, all of their symptoms, while varying from person to person, were relatively mild and lasted no longer than a week, but a sense of anxiety lingered — they’d got a taste of what was in store for tens of thousands of other Canadians.
“To be cleared and resolved, but know the worst was yet to come for Canada, we just had to be part of [the solution],” said Carl.
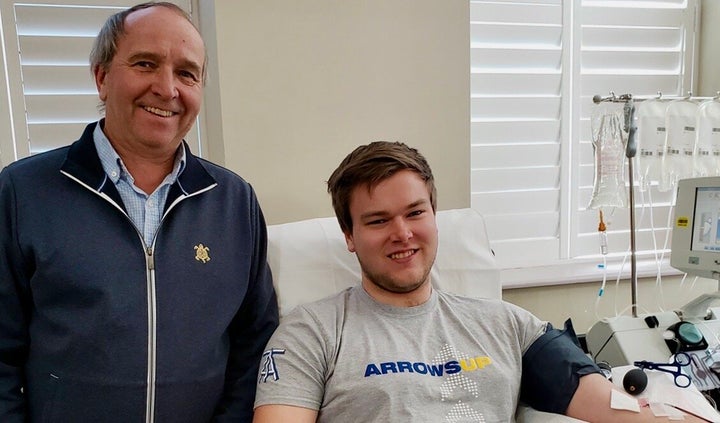
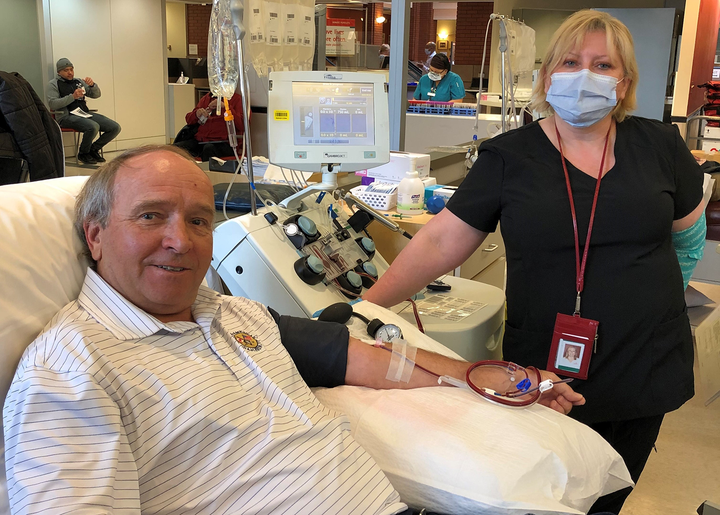
Earlier this month, he and his son Patrick were among the first Canadians to donate their plasma (the liquid part of their blood) to one of the largest clinical trials in the country, involving 11 research teams and 50 hospitals. Canadian Blood Services, Hema-Quebec and New York Blood Centre are collecting plasma from participants.
The researchers are examining if COVID-19 patients benefit from a transfusion of plasma from those who’ve recovered from COVID-19 and built up antibodies that could help fight the disease.
So far, more than 1,000 people have offered to donate their plasma, reported Canadian Blood Services. It’s looking for participants who are younger than 67 years old, have previously tested positive for COVID-19, and have been free of the virus and symptoms for at least 28 days.
“It makes me feel like I’ve made a much-needed contribution,” Carl said.
The plasma trial is an example of a slew of other research and developments taking place across Canada to help end the pandemic.
The race to learn more
Researchers in every province and territory are partaking in national and global trials to determine ways to treat and prevent the coronavirus — options that if proved effective could also be approved by Health Canada to help combat COVID-19.
“I’ve never seen this pace [of research],” Dr. Ramy Saleh, an oncologist who also conducts clinical trials, told HuffPost. “The true word of collaboration is taking place right now, which is great.”
WATCH: How do COVID-19 tests work? Story continues below.
Last month, Saleh launched the website COVID Trials to overcome the “achilles heel” of medical research — recruiting patients. Canadians can directly register to participate in studies on the site, rather than through a physician referral. “The most important part of trials is to recruit, and with COVID-19, time is the greatest enemy,” said Saleh.
Saleh said researchers are looking for a wide range of test subjects — people who’ve tested positive or negative for COVID-19, have been exposed to someone with COVID-19, have experienced mild or severe symptoms, or have recovered. It’s likely to be only a matter of months before they begin enrolling vaccine trial participants.
Chinese company CanSino Biologics has developed a potential COVID-19 vaccine and is in the process of applying to Health Canada to start clinical trials, said the National Research Council of Canada. If approved, clinical trials will roll out this year, carried out by the Canadian Center for Vaccinology in Halifax.
Another study is looking at how effective the arthritis drug Tocilizumab is at reducing inflammation in patients experiencing severe COVID-19 symptoms. Other researchers will look at the impact of treating early cases of COVID-19 with anti-malaria drug hydroxychloroquine.
Quebec researchers are looking at the mental health consequences of social distancing, and in New Brunswick, researchers are studying how people’s health behaviour has changed during the pandemic.
“Without their volunteering or participation, we can never complete those trials and find a treatment or cure for COVID-19,” said Saleh.
Testing for COVID-19 immunity
The need for test participants is only going to grow.
In an effort to screen Canadians for COVID-19 immunity, Health Canada has approved a diagnostic test that detects antibodies. It said in a statement Tuesday, that it aims to test one million blood samples over the next two years.

The serological test, LIAISON, developed by the Italian biotech company DiaSorin, will help health experts determine if people previously infected with the disease can contract it a second time, or if their immune system produces blood proteins to fight it off, as it does for many other viruses, Health Canada said.
The test will also help reveal how widespread the coronavirus is among Canadians generally, and in specific groups that face a greater chance of contracting COVID-19, like health-care workers and the elderly.
“Understanding the scope and scale of COVID-19 infections across the country is key to managing the epidemic over the coming months,” said Health Canada. “This understanding will help inform public health decisions to protect the health of Canadians.”