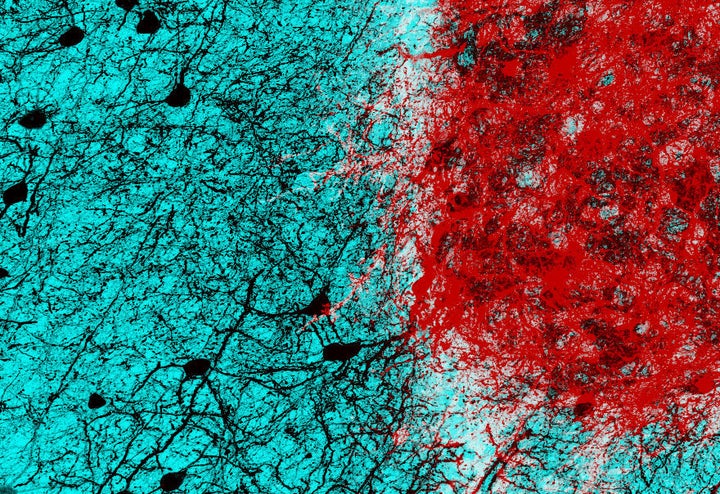
Scientists have long been working toward a day when a traumatic injury or stroke doesn’t cause brain cells to be permanently lost.
Executing this extremely difficult task would involve figuring out how to transplant new neurons into brain tissue. But neurons form precise connections with each other, and are guided by physiological signals that are active during early brain development ― meaning that you can’t sow a fistful of new neurons into mature brain tissue and expect them to grow the way they should.
But scientists are making progress.
Embryonic neurons transplanted into the damaged brain of mice formed proper connections with their neighbors and restored function, researchers wrote in a study published Wednesday in the journal Nature.
By the fourth week, the transplanted young cells became the type of cells normally seen in that area of the brain. They were functional and responded to visual signals from the eyes. Moreover, the cells didn’t develop aberrant connections, something that could lead to epileptic seizures.
“What we did there is proof of concept,” said neuroscientist Magdalena Götz of Ludwig-Maximilians University and the Institute of Stem Cell Research at the Helmholtz Center in Munich, Germany.
“We took the best type of neurons, chosen at a specific time, and then we put them in the lesioned brain,” she said. “That was to find out how well can it work.”
The finding is an important step forward for someday repairing brain injury by using replacement neurons, other researchers said. Still, there are many challenges left.
“I’m excited about this study,” said Sunil Gandhi of University of California, Irvine, who wasn’t involved with the research. “This is evidence that the brain can accept the addition of new neurons, which normally doesn’t happen. That’s very exciting for its potential for cell-based repair for brain.”
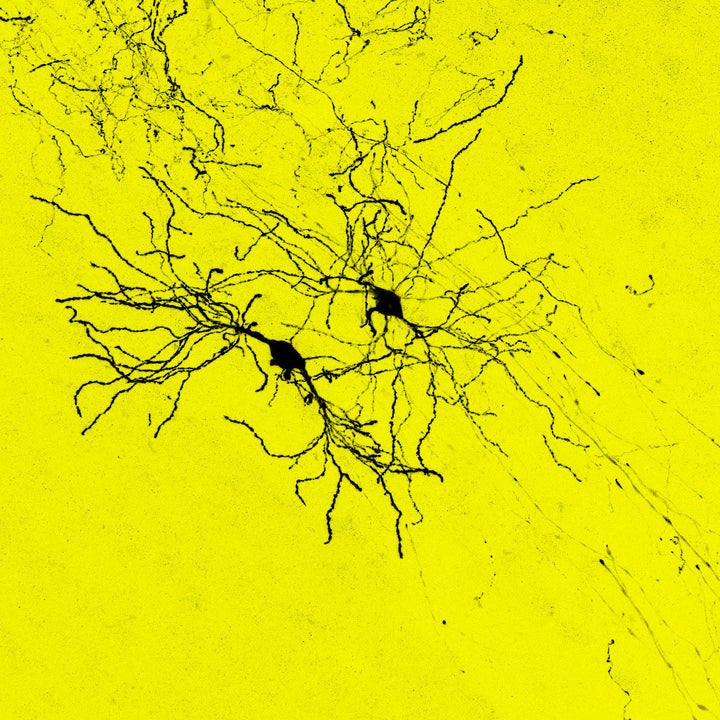
But with complicated human biology comes complicated questions. What if the new cells become cancerous? What if the trauma of brain surgery causes more harm than the good a transplant might bring?
“In the case of stroke, there are therapeutic avenues that involve behavioral rehabilitation that can help to some degree,” Gandhi said. “It is true that the options are limited and frustrating. But the alternative is that we may end up going too fast and have unwanted harmful side effects.”
Neuroscientist Zhiping Pang, of Rutgers Robert Wood Johnson Medical School, agreed.
“This is absolutely an interesting and exciting paper,” he said. “Nevertheless, translating this to human stroke patients, safety will be a concern. A lot more work needs to be done, like the current study, before we can realize this exciting cell-replacement strategy in restoring proper brain functions of a stroke patient.”
The new study is promising, Götz said, but acknowledged that things are a lot messier outside the lab. Injuries to the brain are not clean-cut. They can occur in various sites, involve different types of neurons, and are accompanied by inflammation and other meddling signals. But Götz is hopeful that these problems can be solved.
“We are doing this now in more realistic models, in models of traumatic and ischemic brain injury and all I can say is that it looks pretty good,” she said.
Another challenge is to account for glial cells in the brain, which form scar tissue when an injury happens. That’s why Götz and her team are exploring the potential for turning these glial cells into new neurons that can replace the lost ones.
That approach could also solve the problem of supply, as using cells from fetuses is not a practical option for human patients.
Some forms of neuron transplantation have been done before. People with Parkinson’s disease suffer from a death of dopamine-producing cells deep in the brain, and it’s possible to transplant into their brains new neurons that secrete dopamine and help with certain symptoms. These neurons, however, don’t need to become a part of the existing circuitry. They don’t even need to be human cells ― the first transplant of this kind was done using brain cells from pigs.
Other groups have turned to induced pluripotent stem cells, or adults cells ― from a patient’s skin, for example ― that can be reprogrammed to an embryonic state and then directed to grow into a desired type of neuron.
“What’s going to be important now is to demonstrate that neurons that are grown from pluripotent stem cells can be coaxed to wiring up into the brain,” Gandhi said.
