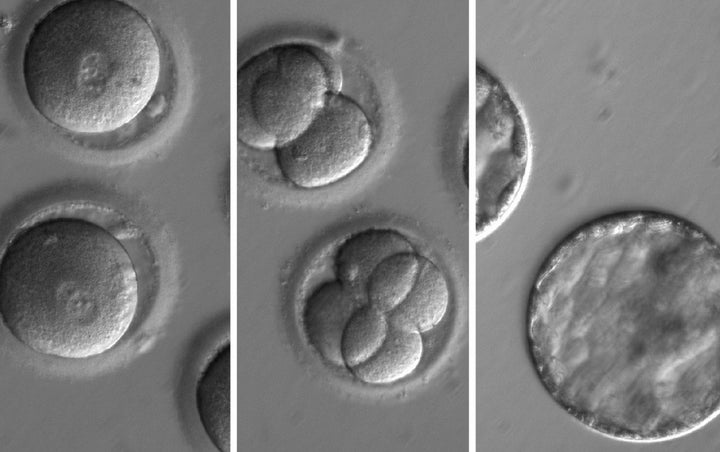
In a breakthrough announced Wednesday, scientists successfully edited the genes of a human embryo to eliminate a dangerous gene mutation. This hasn’t yet produced the birth of an actual baby with genes selected by technological means, but that is on the way — and soon. Science and technological capacity are racing ahead of ethics, safety regulations and our understanding of risks and societal implications.
The potential for “designer babies” has been circulating for years, not just in science fiction but in exciting new research and real-world effects. Already, in-vitro fertilization offers parents assistance in conception and, together with other technologies, allows parents to select healthy embryos, choose the sex of their children and manage a variety of aspects of human reproduction once thought simply to be matters of nature or divine action.
Sex selection, made possible by cruder technologies, has remade the marriage and life prospects of a generation of young Chinese, Indians and others. Now, these capacities have been taken to a new level by advances in technology that allows scientists to manipulate individual genes. The technology is called CRISPR-Cas9 (CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats). The power of this approach was already clear in 2015 when Science magazine named it breakthrough of the year.
What makes the new research just announced another breakthrough is that it is a demonstration that this technique can safely remedy defective (mutated) genetic makeup, producing embryos in which cells are free of the mutation and no new, unwanted mutations are introduced. The specific mutation that was eliminated caused a dangerous heart condition. But there are some 10,000 potentially problematic medical conditions known to be caused by specific inherited mutations. The benefits of successful genetic engineering could be enormous and the attractions to potential parents are obvious.
“Will the new technology only be available to parents in rich and scientifically advanced countries?”
Use of CRISPR-Cas9 technology in “real life,” as opposed to a research setting, is currently forbidden in the U.S. However, genetic modification of other organisms has been permitted (albeit within a complex regulatory regime) for several years, sparking controversies over genetically modified organisms and foods. Researchers can manipulate real human embryos at the point of fertilization but not implant them or facilitate their growth into viable fetuses. The main objection to this — until the new research — has been safety. This has centered on the worries that fetuses would not prove viable or would suffer new mutations.
A recent National Academy of Sciences report suggested that once these safety concerns were allayed, then clinical — that is, real life — use of the genetic engineering techniques should be allowed. It is already allowed in China, and some U.S. researchers worry this is giving the Chinese an unfair advantage. China is also backing some claims to patents for CRISPR-Cas9 while others are litigated in U.S. courts, with implications regarding who can control the technology and its future use.
But safety, in the sense of the well-being of the embryo and potential fetus, is not the only concern. Religious leaders have raised objections over physicians and others “playing God.” The Catholic Church insists that human beings are — or must be — “begotten, not made.” But there are concerns not only about the ultimate questions of the nature and meaning of life. There is also worry that IVF raises challenging ethical questions for young men and women who donate sperm and eggs, without adequate care for what it will mean to them to be anonymous, undisclosed parents. Pope Francis called artificial reproduction technologies “a false compassion.”
Secular thinkers also worry that technology is giving some decision makers a degree of control over human lives, and indeed the human future for which they are not adequately prepared. Are doctors prepared to help make fundamental ethical choices about which babies should receive which benefits — or to assess risks? Are prospective parents? The premier bioethics research organization, the Hastings Center, asks: “What does it mean to be a good parent in the genomic age?” As children are genetically altered, what may this mean for “natural” family bonds?
“Governments could engineer children to win Olympic medals, be better soldiers, be compliant workers or be brilliant scientists.”
It is hard to object to sparing newborn children the burden of inherited diseases. But consider the question: Which newborn children should get that benefit? Is it to be distributed on the basis of parents’ ability to pay? Should it be distributed by the state on the basis of some combination of considerations, like who is likely to be useful to the state and who has connections to those in power? Will the new technology only be available to parents in rich and scientifically advanced countries? Whatever the regulations developed in one country, in the absence of international agreements, will parents with enough money simply go to other countries as “medical tourists”? What will be the legal status of genetically modified offspring? Do parents “own” their children, and is this the basis for a right to decide their basic biological makeup?
Any new technique of enormous power raises questions about how to allocate access, about unintended consequences, and about social and moral responsibility. In a democratic country, it demands public debate. We are short of both. In his book Playing God?, the sociologist John H. Evans argues that our public debate is thin partly because we ask only about what techniques are legitimate, not what goals we should embrace. The potential benefits of gene editing do not stop with reducing the risk of heart disease or cancer. Parents could choose genetic modifications intended to boost intelligence, athletic ability or longevity. The social implications of genetic engineering could be dramatic. As is often the case, how big the impacts are and whether benefits exceed costs and damages depend on how the technologies are used.
Genetic modification produces inheritable differences among human beings. Parents could make their children taller or blonder, more often male, or less likely to get cancer. Governments could engineer children to win Olympic medals, be better soldiers, be compliant workers or be brilliant scientists. Could this reinforce old racial divisions or create new ones? Could this be, as the sociologist Troy Duster put it, the “backdoor to eugenics”?
“Could this be, in the phrase of Troy Duster, the 'backdoor to eugenics'?”
Genetic modification challenges our very idea of human nature. It suggests that we can make human beings into what we want them to be. That’s a little less new than it sounds. Humanity has already been reshaped by the development of language, literacy and education. It has been reshaped by better nutrition and sanitation, with enormously positive consequences for human life expectancy. But the capacity to consciously alter human beings gene by gene is new.
What does this mean for the notion that all human beings are members of a single species and thus members of a common “community of fate”? This idea is basic to the notion of human rights. It is basic to the way citizenship is understood in most countries. Is it possible that genetic engineering could create such marked differences about human beings that they couldn’t all be considered citizens even if they all descended from people who are citizens today?
Gene editing is one of the most promising medical technologies in years. But unless there is much more attention to the ethical and social choices before us, we risk seeing that promise mired in controversy — or turned into a disaster.

