Let's say you have developed a great prototype, but the prototype needs to get to market. It needs to get to commercialization. Commercialization is key to impact, but we have a real shortage of groups that will support the "unsexy" part of product development - scaling. My plea is - particularly to funders - to focus support on the valley of death between proof-of-concept and commercialization. Without it, there is no impact at scale.
D-Rev is a non-profit social enterprise. Our innovations are medical technologies that close the quality healthcare gap for underserved populations. There are more than 4 billion people in the world living in poverty. In many cases they don't have access to clean water, to health products, to all sorts of basic items that can improve their lives. And with good products, they can be more productive; their children can go to school; they can better take care of their families and have jobs outside their home.
There are three things to know about D-Rev's products. First, our products are world-class. This means that they perform on-par or better than the best products in the market. Products serving the poor tend to be low-tech or low-quality, but at D-Rev we say everyone deserves great design; you can design the very best products for poor people. Second, our products are market-driven. Third, they are user-centric. This means not just designing for the end-user, but designing for everyone in that customer chain, from the purchasing decision maker to the repair technician.
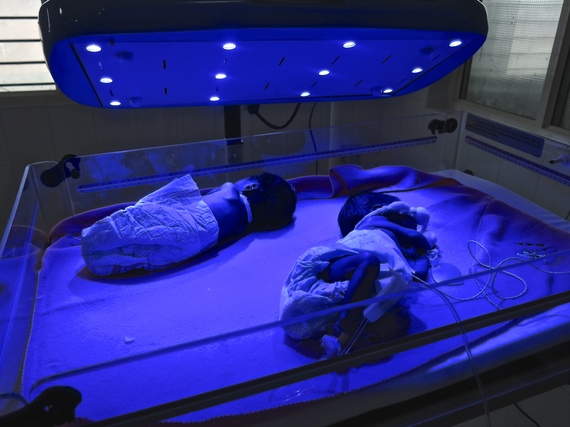
Our Brilliance project started when an Indian doctor approached one of my colleagues and said, "Everyone is focused on the big-ticket items in neonatal health, but there's low-hanging fruit. There are babies who are literally dying because they don't have treatment for jaundice."
Brilliance is a phototherapy device, which is blue light used to treat severely jaundiced babies. We licensed it to the largest maker of neonatal equipment in India, Phoenix Medical Systems, and we fixed the price so that there is price-transparency, which is very rare with medical devices. Because we are not just focused on our users, but on increasing impact, we also agreed to take lower royalties from sales to public or rural hospitals. Brilliance has now been sold to 37 countries and, based on confirmed installations, has treated approximately 100,000 babies in its first three years on the market who otherwise would not have received effective treatment.
Another product we've designed is the ReMotion Knee, which is a prosthetic knee for above-knee amputees. The ReMotion Knee launched in December 2015.
We think about the entire product lifecycle - not just the design of the product, but the problem we're trying to solve, and how we achieve impact. It's a four-step process.
The first step is Identify, which is about understanding what the problem is, who the users are, what the markets are, and what the price point needs to be.
The second step is Design. Design is what you traditionally think of with product development. It is brainstorming around the crazy, far-out ways we might be able to solve this problem. It's prototyping, iterating, and talking to users to get to a final embodiment of the product that will go through manufacturing and commercialization.
The third step is Deliver, and this is manufacturing, distribution, marketing, and sales. If you are designing medical devices, delivering also includes regulatory approvals. At D-Rev, we think about the delivery phase as early as possible in the design process.
The fourth step is Impact Assessment. Impact assessment helps to determine if your product is, in fact, solving the problem you set out to solve. At the end of the day, you want to make sure your target users are using your product correctly so that you are creating impact - and that needs to be measured.
Wherever possible, we design impact assessment into the products themselves, and make the results transparent and accessible. Brilliance, our phototherapy device, self-tracks. This way we know from the device how much it is used, which enables us to calculate the number of babies treated by Brilliance units. From those numbers we can estimate the numbers of babies who otherwise would not have received effective phototherapy (the counterfactual), and the number of deaths and disabilities averted. These data are displayed on our Impact Dashboards.
One of the things we recognized very early on at D-Rev is that we can't do everything, particularly around delivering products. To solve this problem, we work with all kinds of partners. We work with NGOs, for-profit companies, and faith-based organizations. What we are looking for in our partnerships is mission alignment - two aligned organizations can have more than simply double the impact. That means getting the products to not just users, but doctors and patients who otherwise would not have access to quality health technologies.
A great example of that is Phoenix Medical Systems, our Indian partner for Brilliance. Phoenix is a for-profit and D-Rev is a non-profit, but this partnership works because it achieves Phoenix's goals and D-Rev's mission at the same time. Once a D-Rev product goes on the market, it needs to be entirely economically self-sustaining. Because we use the private market to deliver our products, we need to design not just a very affordable product, but an affordable product that has profit margins built-in, so that if D-Rev exits the product, the product will continue to be sold and have impact due to a financial incentive structure in place.
We are structured like this because we think it is the best way to scale our products. Responding to the market also holds us accountable to our users and customers - ensuring that we are designing a product that solves a real problem. When you have partners that are working together to understand users' needs and develop a solution, when you align incentives and mission, that's how you design products for impact.
This essay is an updated version of one published in December 2015 on the World Economic Forum's blog.