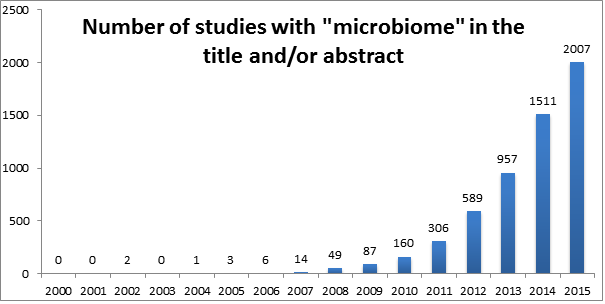
If you ask the average person what they know about the human microbiome, they probably won’t have much to tell you. You can’t blame them though, as can be seen from the figure below, the number of scientific papers on the microbiome has surged only recently.
https://biosciencephdforum.wordpress.com/2016/02/18/a-crappy-phd-my-journey-into-microbiome-research/
So what exactly is the human microbiome? The human microbiome is a collection of microorganisms - bacteria, viruses, fungi and archaea throughout the human body. They’re on your skin, in your lungs, and in your gut. The bacteria, which have been the main focus so far of human microbiome research, can be further subdivided into three categories: commensal, symbiotic and pathogenic. Commensal bacteria are bacteria that benefit from the human body but offer no benefit to us. Symbiotic bacteria benefit from living inside of us and are good for our health. Pathogenic bacteria are those that are capable of causing disease. With these three types taken together, a crude estimate is that there are 10 trillions of these microorganisms in your body, meaning there are 10x more of these microorganisms than there are human cells. The bacteria living in the microbiome protect us from harm by boosting our immune system, while also being the cause of inflammation, tuberculosis and pneumonia.
One of the main things that held microbiome research back several years ago was the high cost of DNA sequencing. As the cost of sequencing decreased, there was an increase of data mining activity to better understand the genomic composition of the microbiome. The end result was larger datasets that researchers could use to extrapolate results from and publish papers with. Unfortunately since this is still a nascent field, there is a lot of room for improvement. Primarily the lack of reproducible papers and discrepancies from diagnostics companies means standards must be set in place for research to be more impactful.
I had a chance to talk with Xavier Duportet, the CEO of Eligo Bioscience a company on the frontier of helping protect our microbiome from pathogenic bacteria. Duportet, who earned his PhD in Synthetic Biology while at INRIA and MIT has raised $3 million so far to help treat and sculpt the microbiome. His company's thesis is that our current approach with antibiotics is a crude method that can cause unintended, dangerous consequences and that a more elegant solution is needed.
The response from the company is to use a synthetic biology platform that combines a nuclease (i.e. CRISPR/Cas system) with protein-based nano-delivery vectors (engineered phage capsids) to develop what they’ve dubbed “eligobiotics”, a new generation of highly precise anti microbials. As opposed to antibiotics which destroy bacteria regardless of whether they are symbiotic or pathogenic, eligobiotics can be programmed to eliminate bacteria based on their genetic sequence. This precision is similar to having a poison that only wipes out a small part of the bacterial population, as opposed to antibiotics, which not only wipe out pathogenic bacteria but also harms symbiotic bacteria essential for your health.
If Eligo prove themselves in clinical trials, it’ll be as important as the discovery of Penicillin - Christian Brechot, President of Pasteur Institute
Duportet shared his enthusiasm for the new technology being developed at Eligo Bioscience, which has been published in major peer-reviewed journals (Nature Biotechnology) and which has already been shown to efficiently and precisely eradicate highly pathogenic and antibiotic-resistant Staphylococcus aureus and Escherichia coli bacteria. Early research has shown that eligobiotics work in petri dishes and also in more complex animal models, which gives serious hopes for future development.

Duportet explained to me that his company, like many biotech companies, started with a research paper and the support of professors. In fact when Duportet brought the results of the Rockefeller team with whom he was working with to a close professor at MIT working next door to his lab, they realized that they happened to both be working on similar technologies, and as a result decided to join forces. The Paris-based company, which has attracted other scientists from MIT, Stanford, Inserm and Institut Pasteur, as well as former executives from large big pharma, is now about to raise their Series A round. Their goal is to push the technology to the clinic and test it in humans in the next two years. Duportet hopes to first create efficient and targeted therapies to treat highly deadly bacterial diseases in children and elderly people, and then later on focus on targeting more complex diseases that are beginning to be associated with the microbiome.
For a long time, the scientific community has for a long time been warning the world of “superbugs”, bacteria that have developed a resistance to our antibiotics. The rise of “superbugs” is indeed becoming one of the most worrying threats for the population globally, with worrisome predictions from the World Health Organization that infections caused by antibiotic resistant bacteria will be the main cause of deaths in 2050, with more than 10 million people dying from antibiotic resistant infections per year.
While the path to regulatory approval for drugs is a long one, Duportet is confident that the company's highly precise antimicrobials will be pivotal not only in battling “superbugs”, but also by helping keep our microbiome healthy.
