You’ve probably heard of people having filler injected into their faces for a fuller, more youthful appearance, but did you know people also get filler injected into the backs of their hands? They do. And now it’s FDA approved.
The Food and Drug Administration recently approved Restylane Lyft, a hyaluronic acid filler, for use in the hands; it’s the first and only hyaluronic acid injectable gel to be FDA approved for an area other than the face. (Radiesse, a dermal filler of calcium hydroxylapatite, has also been approved by the government for use in the face and hands.)
“Filler in the hands is used like filler in other places, to cause a rejuvenating effect by giving more volume and plumpness to an area which has lost some of that,” Dr. Michelle Yagoda, a New York-based facial plastic surgeon, told HuffPost. “With aging, there’s a loss of collagen and elastin, and the skin gets thinner, and in the hand, when that happens, you can see the tendons and the veins in the hands more prominently, and people associate that with aging.”
By using filler, Yagoda added, you can plump the back of the hand to make the tendons and veins less visible. People tend to get fillers when they think the appearance of their hands doesn’t match their face, “giving their age away,” Yagoda said.
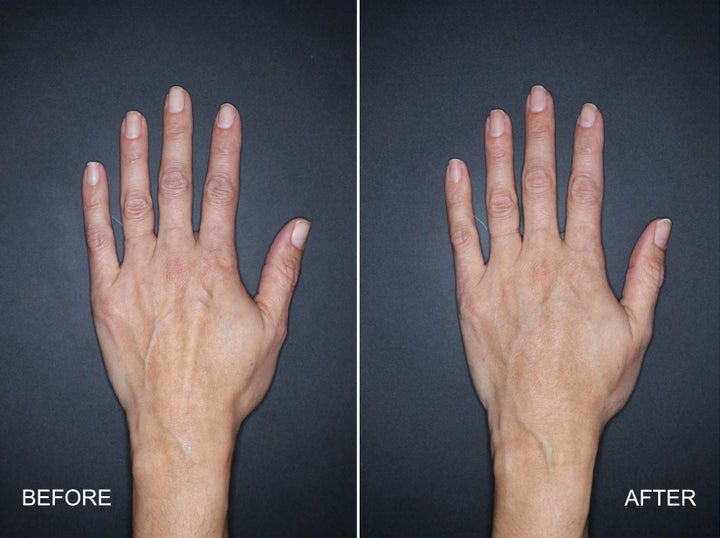
Filler in the hands definitely isn’t new.
“Fillers have been used off-label in many areas, including the hands, for a long time,” Dr. Ellen Marmur, a board-certified dermatologist in New York City, told HuffPost, adding, “aging hands [are] definitely one area that bothers a lot of people more than you might think.”
Doctors have also been able to use a patient’s own fat to fill in the loss of volume on the hands, Dr. Stafford Broumand, a New York City-based plastic surgeon at 740 Park Plastic Surgery told HuffPost. Keeping the hands looking youthful, Broumand added, is part of the “journey of trying to look better as they’re aging.”
“If you don’t address all the visible parts or all areas of the body, you’re going to be left behind in one spot,” Broumand said.
Marmur said that some people used to do sclerotherapy, a procedure that involves injecting a salt solution into the veins to essentially get rid of them. But, she said, “you kind of need those veins.”
How does the filler work?
Different providers administer fillers in different ways. Some doctors, Yagoda said, make multiple injections in small amounts between the tendons, while others may make one large injection in the center of the hand and then move the filler to where it needs to go once it’s under the skin.
“In either case,” she said, “the skin is numbed up, either with a topical anesthetic and/or a topical anesthetic and ice. [Then] the skin is tented up ― pinched up ― and the injection is delivered right into that area of the skin above the tendons and muscles in the hands.”
Broumand noted that some swelling or bruising might occur, but as long as the hand is numbed properly, it shouldn’t be painful or uncomfortable.
The filler could last nine months to a maximum of two years, Marmur said, while Yagoda said it typically lasts six to 10 months.
Broumand noted that fillers usually dissolve after about a year, so regular touch-ups can help maintain the look.
And if you’re wondering how much this might cost, you can expect to spend a couple of thousand dollars for the initial session, Yagoda and Broumand said.
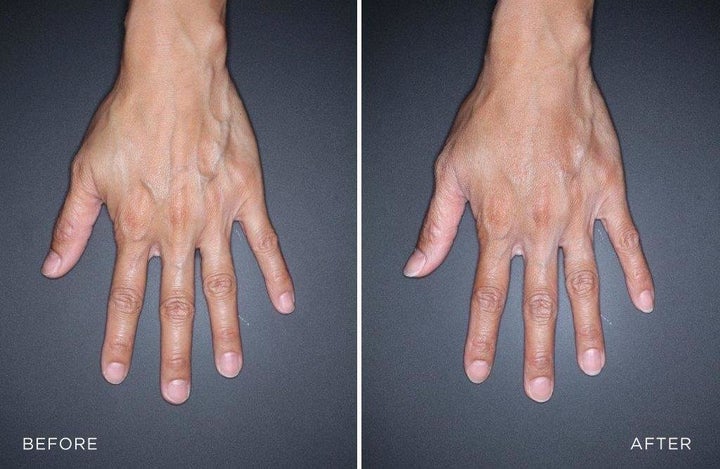
So what’s the big deal with FDA approval?
The doctors we spoke to agreed that FDA approval for Restylane Lyft in the hands may change a few things regarding the treatment and how it’s viewed by those in the cosmetic surgery field.
“For patients and for doctors,” Marmur said, “it’s wonderful for us to have the reassurance that we have FDA approval for the hands, so we know it’s safe and effective.”
With FDA approvals, Marmur said, come better safety standards and record keeping.
Since the use of fillers in the hand has been largely off-label, Broumand said, there has been no public discourse on it. If there isn’t FDA approval for use in a specific area, a company can’t even talk about it, he said. Additionally, if a filler is being used off-label, the manufacturer can’t train doctors on how to use it that way, nor can it provide information about off-label uses, Yagoda added.
Now there will be more discussion and awareness of options available “to treat the aging hand and to keep it on par with the rest of the face and body,” Broumand said. “This gives it a broader sounding board for people to talk about it and for people to showcase the advantages for the top of the hand.”
Yagoda also added that the FDA approval may encourage more physicians to administer the fillers in hands.
“There is always a small group of providers that don’t use things in an off-label manner,” she said. “And those physicians may begin to use the filler in the hands or other areas in the body because they feel that they can get training, they can ask questions about it or because they feel it’s safe enough.”
For anyone considering getting fillers in their hands, Broumand advised: “Go to someone who’s qualified, talented and has the best interests of the patient at heart.”