Cancer is actually more than 200 specifically different conditions that are all characterized by uncontrolled cell growth. Cancers have traditionally been classified by the cell of origin and by the body part where they occur. Today researchers are focusing on the types of DNA mutations that drive cancer growth, the biological processes that various types of cancers have in common, and how understanding those can lead to new treatments.
Can you imagine what we’d learn if we could actually see cancer growing inside a live organism, watch how it happens, and understand why? New technologies now allow this, and important lessons are coming from lab mice bred specifically for cancer research.
The National Cancer Institute (NCI) says that humans have studied patterns of inheritance in mice for over 3,000 years. Beginning with exploring how inheritance patterns affect physical mouse characteristics like coat or eye color, researchers have been studying mice in scientific labs since 1964. Most recently they are trying to understand what biological, physiological, and genetic factors define their (and our) susceptibility or resistance to cancer.

NCI explains that mice as models for research are easy to grow, handle, and reproduce; more importantly they have significant genetic similarities with humans. For those of us who think of mice as pests or nuisances that invade our homes and garages, the idea of mouse as teacher (especially on a topic as confounding as the mechanisms that drive cancer biology, metastases, and responsiveness to treatment) may not be obvious.
Some mouse strains are genetically engineered to develop specific types of cancers, while others receive cancer transplants to reveal how their bodies’ natural processes and blood chemistry react to and are affected by its presence. These preclinical studies reveal important learnings about both cancer genes and other factors within host tissue that accelerate or hinder cancer growth and treatment effects.
A topic of particular focus by scientists who work with mice has been oncogenes (those genes that drive cancer growth) and tumor suppressor genes (those that prevent cancer growth). Scientists can now breed mice to remove (knock out) or replace (knock in) certain segments of mouse DNA in order to understand the development, progression, and treatment of cancers. This research has been particularly powerful regarding breast, lung, brain, skin, and pancreatic cancers in mice and has impacted cancer treatment in people.
Now many studies are investigating various proteins and enzymes in the cancer’s microenvironment and vasculature that affect cancer growth and treatment resistance.
One of the breakthroughs in cancer research was recognition of angiogenesis, the formation of new blood vessels, as a key to cancer progression. This field of research was pioneered by Dr. Judah Folkman from the early 1970s through mouse studies that demonstrated how access to a blood supply fuels tumor growth and how inhibiting the supply curtails it.
Subsequent research in the late 1990s pioneered by Dr. Rakesh Jain, Director of Steele Laboratories at Massachusetts General Hospital (MGH), revealed how the abnormal structure of the newly-formed blood vessels inside tumors may actually impede chemotherapy from reaching the cancer cells. Dr. Dan Duda, who leads a lab at MGH, is one of the scientists further exploring this issue.
Duda’s group uses surgically implanted windows in the bodies of mice to allow repetitive and minimally invasive microscopic-level imaging of the blood vessels in both healthy and cancerous mice. Micrographs (photos through microscopes) of mouse tumors, taken through windows in real time or at frequent time intervals, reveal how the vasculature changes during treatment, how blood cells move through the blood vessels, and whether the body’s immune response has been activated against the tumors. This allows investigators to literally see in real-time how various therapeutic agents affect cancer size and growth rate.
An important building block for Dr. Duda’s work has been the observation that in healthy tissue, blood vessels are largely linear in pattern (as shown in red below), allowing blood and anything carried by the blood to pervade the tissue at a consistent and predictable rate:
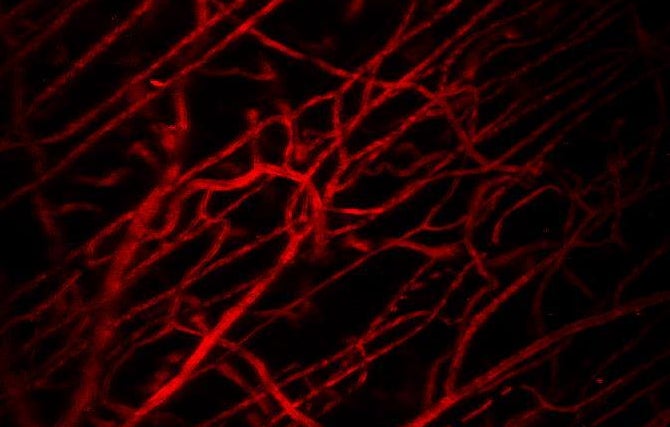
Vascular structure in healthy tissue evaluated by intravital microscopy
The companion observation was that in cancer tumors, the blood vessels are abnormally contorted, as shown in green in the photo below. The kinks and curls, as well as the absence of vessels in some areas, impede blood-borne chemotherapy from reaching the very cancer it is designed to kill:
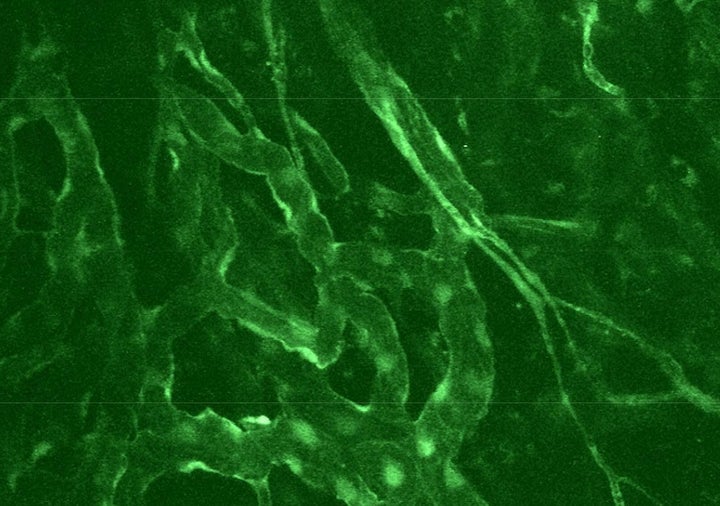
Vascular structure in a brain metastatic breast cancer in a mouse evaluated by intravital microscopy
The lab is now exploring different drugs that may smooth out the contours of the blood vessels to allow better direct transmission of the chemotherapeutics or biologic therapeutics to the cancer cells. Through this approach, investigators seek to understand cancer recurrences and to determine how to make cancer therapies more effective for particular types of patients and cancers.
Research like this, which has the potential to change the game in cancer therapy, takes substantial amounts of time and money. For Dr. Duda’s work, machines and scientific techniques didn’t previously exist to perform the needed imaging and assays to test working hypotheses. Funders for needed inventions had to be engaged before research could progress. Then researchers could seek out clinical markers to help target treatments for individual patients, develop preclinical models that demonstrate the mechanisms by which treatments blunt cancer growth, and conduct clinical trials to prove positive impacts.
Dr. Duda’s lab is collaborating in 37 clinical studies that use molecular, cellular, and imaging analyses to understand the impact of drugs on the immune system in fighting cancer. His staff has quadrupled over the past six years. Yet with substantial reductions in federal funding, significantly more financial support is needed in order to maintain this unique group of scientists and carry their learnings into clinical applications that can improve patient outcomes and save lives.
To support such breakthrough research, go to https://giving.massgeneral.org/donate/ and designate your gift to Dr. Duda’s program.
